 
Owning to the nanometer sized diameter and tunable length which can reach up to a few centimeters CNTs can be a drug vehicle for therapy purposes. has been using CNT, in partnership with Zyvex Performance Materials, in designing bicycle components like handlebars, seatposts, crank etc. Owning to its stiffness, mechanical strength, conductivity and structure carbon nanotubes have widespread applications in the area of sports, transistors, material science, biology etc. 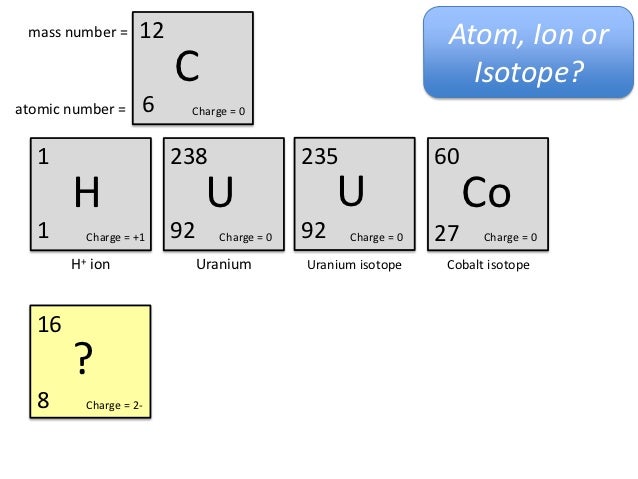
A typical carbon nanotube is show in Figure 1. These bonds are stronger than the alkanes and provide CNTs which extremely high in strength. In more chemical terms, carbon nanotubes bond due to sp2 hybrid carbon atoms. Individual CNTs can align themselves by pi-pi stacking and Van der Waals interactions. Nanotubes can be broadly divided into two types: These graphene sheets are rolled at a specific angle, which decides the properties of CNTs. ĬNTs belong to the fullerene family and the name is derived from their long and hollow structure where walls are formed by carbon atoms sheets called graphene. Owing to this extremely high strength, CNTs have found a place in the materials used for designing bats, car parts etc. In addition to the mechanical strength and stiffness, it has high thermal conductivity and wide range of electrical properties. CNTs have been constructed in a wide range of diameter to length ratio. This material has significantly higher stiffness and exceptional strength. This nanostructure has unusual properties which increases its applications in the field of biology, electronics, water desalination, material science and optics. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.Carbon nanotubes (CNTs) are carbon allotropes having a cylindrical nanostructure. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Elements Common Charges 1 Charge of Hydrogenionġ+ 2 Charge of Heliumion 0 3 Charge of Lithiumionġ- 10 Charge of Neonion 0 11 Charge of Sodiumionġ- 18 Charge of Argonion 0 19 Charge of PotassiumionĢ+, 3+, 4+, 5+ 24 Charges of Chromiumions 2+, 3+,6+ 25 Charges of Manganeseions 2+, 4+, 7+ 26 Charges of Ironionsģ+ 32 Charges of Germaniumions 4-, 2+, 4+ 33 Charges of Arsenicions 3-, 3+, 5+ 34 Charges of Seleniumions 2-, 4+, 6+ 35 Charges of Bromineionsġ-, 1+, 5+ 36 Charge of Kryptonion 0 37 Charge of Rubidiumion 1+ 38 Charge of Strontiumion 2+ 39 Charge of Yttriumion 3+ 40 Charge of Zirconiumion 4+ 41 Charges of Niobiumions 3+, 5+ 42 Charges of Molybdenumions 3+, 6+ 43 Charge of Technetiumion 6+ 44 Charges of Rutheniumions 3+, 4+, 8+ 45 Charge of Rhodiumion 4+ 46 Charges of Palladiumions 2+, 4+ 47 Charge of Silverionġ+ 48 Charge of Cadmiumion 2+ 49 Charge of Indiumion 3+ 50 Charges of Tinions 2+, 4+ 51 Charges of Antimonyions 3-, 3+, 5+ 52 Charges of Telluriumions 2-, 4+, 6+ 53 Charge of Iodineionġ- 54 Charge of Xenonion 0 55 Charge of Cesiumionġ+ 56 Charge of Bariumion 2+ 57 Charge of Lanthanumion 3+ 58 Charges of Ceriumions 3+, 4+ 59 Charge of Praseodymiumion 3+ 60 Charges of Neodymiumions 3+, 4+ 61 Charge of Promethiumion 3+ 62 Charge of Samariumion 3+ 63 Charge of Europiumion 3+ 64 Charge of Gadoliniumion 3+ 65 Charges of Terbiumions 3+, 4+ 66 Charge of Dysprosiumion 3+ 67 Charge of Holmiumion 3+ 68 Charge of Erbiumion 3+ 69 Charge of Thuliumion 3+ 70 Charge of Ytterbiumion 3+ 71 Charge of Lutetiumion 3+ 72 Charge of Hafniumion 4+ 73 Charge of Tantalumion 5+ 74 Charge of Tungstenion 6+ 75 Charges of Rheniumions 2+, 4+, 6+, 7+ 76 Charges of Osmiumions 3+, 4+, 6+, 8+ 77 Charges of Iridiumions 3+, 4+, 6+ 78 Charges of Platinumions 2+, 4+, 6+ 79 Charges of Goldions 1+, 2+, 3+ 80 Charges of Mercuryions 1+, 2+ 81 Charges of Thalliumions 1+, 3+ 82 Charges of Leadions 2+, 4+ 83 Charge of Bismuthion 3+ 84 Charges of Poloniumions 2+, 4+ 85 Charge of Astatineion Unknown 86 Charge of Radonion 0 87 Charge of Franciumion Unknown 88 Charge of Radiumion 2+ 89 Charge of Actiniumion 3+ 90 Charge of Thoriumion 4+ 91 Charge of Protactiniumion 5+ 92 Charges of Uranium ions 3+, 4+, 6+ Free Gift for you: Interactive Periodic Table List of elements with their common ionic charges are mentioned below.Įlements with multiple ionic charges are also mentioned in this table. When atoms gain electron/s, the negatively charged ion is formed, and when the atoms lose electron/s, the positively charged ion is formed. This electric charge generated on the ion is known as Ionic charge. Ionic charge: When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
